Alzheimer's research at ULB
Most age-related neurodegenerative diseases are associated with aberrant accumulations of specific misfolded proteins in the nervous system. Among them, Alzheimer’s disease (AD) is the most frequent neurodegenerative disease leading to dementia and cognitive deficits. AD, thus, represents a major health challenge for our aging societies.
- Neuropathological lesions
-
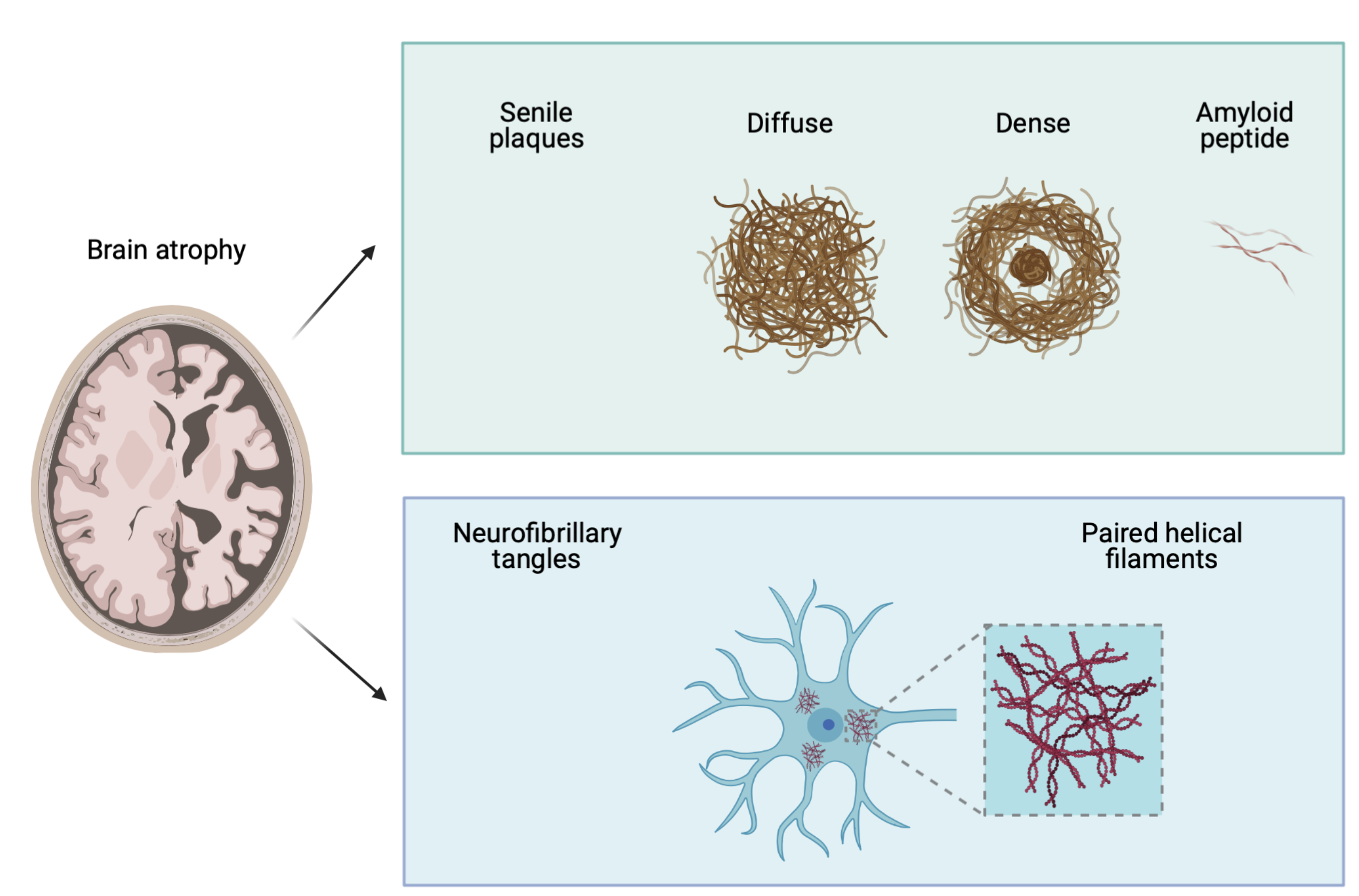
Alzheimer’s disease is a neurodegenerative disease characterized by the presence of two cerebral lesions called senile plaques and neurofibrillary tangles.
Senile plaques are composed of extracellular deposits of amyloid peptide.
Neurofibrillary tangles (NFT) are intraneuronal lesions composed of phosphorylated and aggregated tau proteins forming paired helical filaments. These lesions are highly correlated with neuronal loss and the cognitive decline.
Both lesions are validated as diagnostic and biomarkers of the disease (e.g. by neuroimaging and in the cerebrospinal fluid). Due to our limited knowledge of fundamental cellular mechanisms of the disease, there is yet no disease-modifying drug available for this devastating disease .
- Tau pathology formation and propagation
-
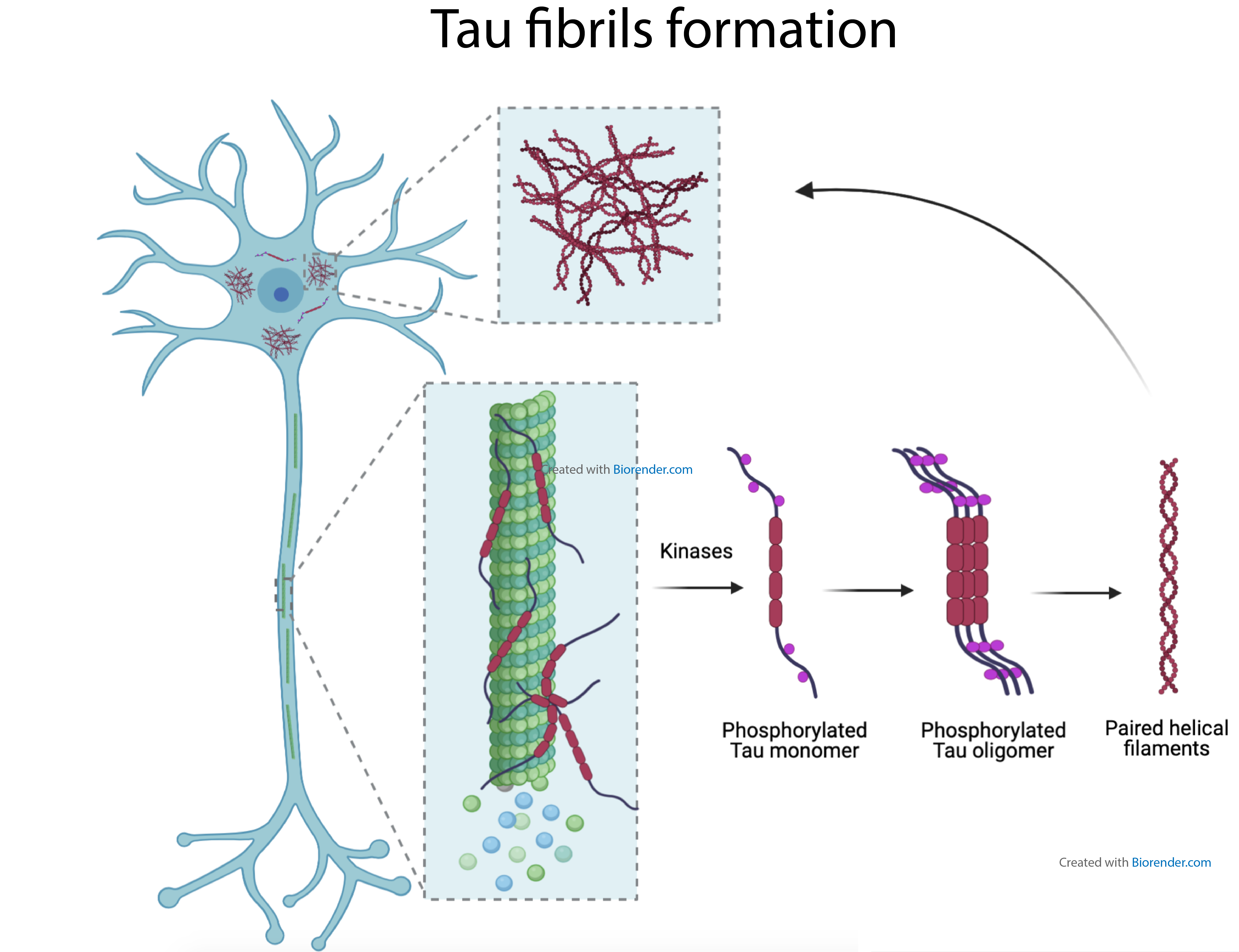
Neurofibrillary tangles (NFT) are composed of intraneuronal aggregates of tau proteins forming paired helical filaments (PHF). The progressive development of tau pathology in brains of Alzheimer’s disease (AD) patients is neuropathologically ranked into 6 stages according to the neuroanatomical spreading of NFT. Interestingly, NFT do not develop randomly in the brain during the progression of disease, but their spreading occurs in a stereotypical fashion across neuroanatomically connected areas. This suggests that pathological tau “seeds” can be transmitted via synaptically connected neurons in a prion-like manner, inducing tau aggregation by the recruitment of normal tau by abnormal tau seeds.
In AD brains, tau undergoes several post-translational modifications such as hyper- and abnormal- phosphorylation, cleavage and pathological conformational change. This abnormal and hyper-phosphorylation of tau seems necessary for the formation of fibrillary tau proteins aggregates. Hyperphosphorylation of tau protein leads to tau detachment from microtubules and to tau accumulation in neuronal cell bodies. Cytoplasmic tau accumulation can be observed even at an early and pretangle stage preceding the formation of the neurofibrillary tangles.
- Our approach
-
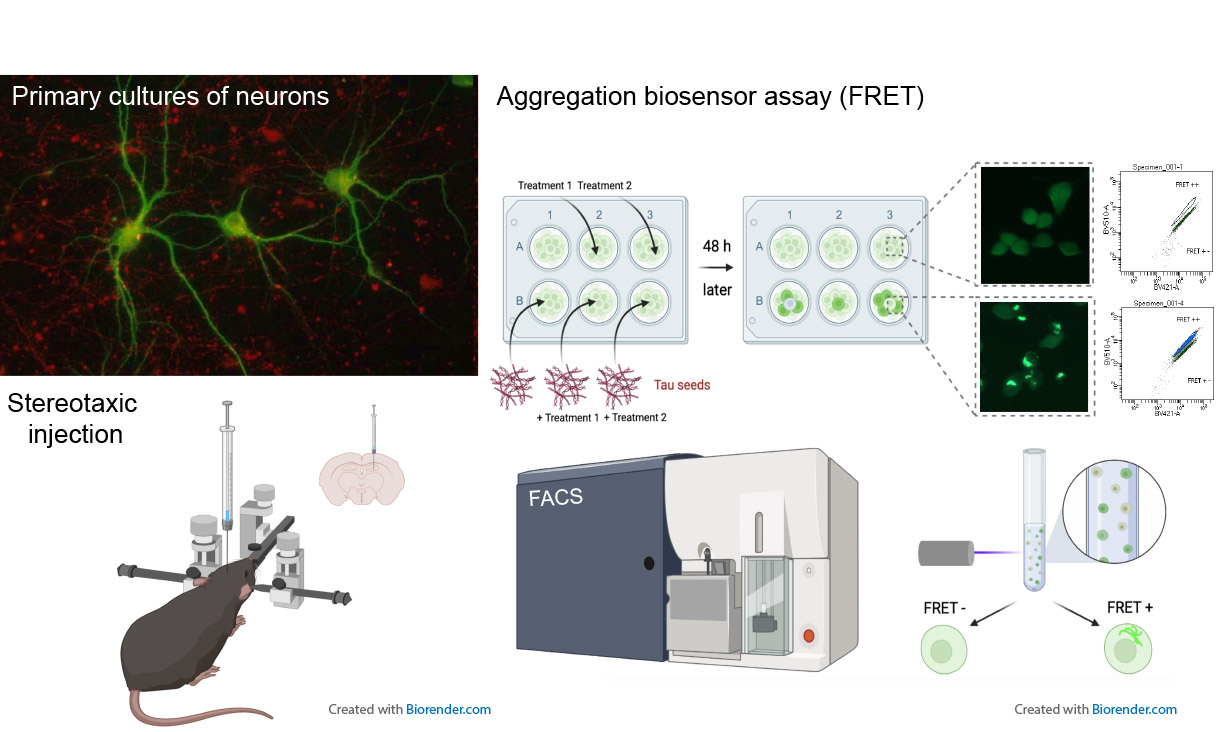
My field of research is devoted to the understanding of the mechanisms of formation of neurofibrillary tangles by analysis of human pathological brain tissue, cellular and animal models and to the preclinical therapeutic trials against AD and related tauopathies.
Université Libre de Bruxelles

ULB Neuroscience Institute

Fondation Recherche Alzheimer

FNRS